 |
Sulfer |
 |
Physiological Role
Metabolism of Sulfur
I. Physiological Role
- Component of sulfur amino acids – methionine, cystine, cysteine, homocyteine, cystathione
- Oxidation-reduction reactions
- The disulfide bond is easily converted to sulfhydryl groups and vice versa
+2H
Cystine 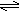 2 Cysteine
2 Cysteine
-2H
- Biologically active compounds activity is dependent upon oxidation or reduction of SH groups:
- Insulin
- Glutathione (oxidation-reduction reactions; transport of amino acids)
- Coenzyme A (transport of acyl groups)
- Lipoic acid (one of coenzymes participating oxidation of a-ketoglutarate to succynyl CoA)
- Water soluble vitamins thiamin and biotin contain sulfur
- Component of the structural protein collagen
- Other important sulfur-containing materials
- Mucoitin sulfates (protect GI walls against digestive enzymes)
- Heparin (blood anti-coagulant in lung and artery walls)
- Fibrinogen (fibrin is framework for blood clot)
- Hemoglobin (transport of O2 and CO2 in blood)
- The cytochromes (electron transport, oxidative phosphorylation, detoxification or xenobiotics)
- Sulfates detoxify by forming esters
II. Metabolism of Sulfur
- Ruminants
- Rumen microorganisms use inorganic sulfur to synthesize sulfur amino acids and vitamins
- Sulfate is reduced to sulfite by rumen bacteria
- Bacteria incorporate reduced sulfur into organic compounds
- Much of consumed S is converted to cysteine and methionine
- Organic S compounds are absorbed when bacteria are digested
- Large quantities of sulfide are also absorbed from the rumen
- Endogenous S is recycled via saliva to the rumen
- Sulfur from metabolism of S containing compounds is also excreted in the urine
- Nonruminants
- Only small amounts of inorganic S are usually present in diets of nonruminants
- Sulfur is largely ingested and absorbed as organic compounds
- Methionine, biotin, and thiamin are the only sulfur compounds required in the diet
- All other S compounds can be synthesized from products of methionine catabolism
- Absorption of sulfur
- Both inorganic and organic forms of S are absorbed by active transport from the small intestine
- Absorption of sulfate is usually 90% or greater
- Excretion of sulfur
- Inorganic sulfate (SO4)
- Neutral S (cystine, taurine, thiosulfates)
- Esterifed S (complex detoxification products)
- Interactions of sulfur with other elements
- Orbital structures are similar for S and Se
- Added dietary sulfate increases requirement for Se
- Se can replace S in methionine and cysteine
- Sulfate reduces toxic effects of excess Se
- S-Cu-Mo interaction will be discussed later
- Recommended allowance for sulfur:
- Ruminants: 0.1% - 0.2% in dietary DM
- Nonruminants: 0.3% - 0.8% sulfur amino acids in dietary DM
- Sources of sulfur
- Sulfur deficiency
- Ruminants
- Inadequate S nutrition of microbes
- Depressed diet digestibility
- Depressed microbial protein synthesis
- Suboptimal utilization of nonprotein nitrogen
- Reduced numbers of rumen microbes
- Effects on host animal
- Negative nitrogen balance
- Excessive tearing and salivation
- Dullness, emaciation, weakness
Nonruminants
- Deficiencies of organic molecules that contain sulfur produce symptoms characteristic of the specific molecule that is deficient
- Sulfur excess
- Ruminants (J Dairy Sci. 67:2179, 1984)
- 0.4% S as sodium sulfate is believed to be maximum tolerable dietary S
- Causes of S toxicity in ruminants
- Use of gypsum to limit feed intake
- Use of ammonium sulfate as a major source of nonprotein nitrogen or to decrease cation-anion difference
- Clinical signs of excess S
- Reduction in feed intake, ruminal digestion, rumen motility, and rumination
- Twitching of jaw, eyelid, and ear muscles
- Evidence of pain
- Staggery gait
- Severe diarrhea
- Advanced stages of S toxicity
– Inability to rise
– Grunting and fast, labored breathing
– Coma and death
- Causes of death
– Acute mortality: Action of hydrogen sulfide on respiratory and central nervous system
– Later mortality: Dehydration from diarrhea
- Nonruminants
- May be less tolerant of sulfide than are ruminants. Maximum tolerable level may be 0.2% - 0.3% dietary inorganic S
- Animals over liquid manure pits may be exposed to sulfur gases
- Effects on swine of inhaling air containing different amounts of hydrogen sulfide:
- 0 – 9 ppm.........None
- 5 – 10 ppm.......Temporary eye irritation, nasal discharge, shallow breathing
- 28 ppm.............Loss of sense of smell
- 47 ppm.............Discomfort
- 180 ppm...........Severe toxicity
- 447 ppm...........Death

 For individual consultation or questions about our products, call
1-800-628-0997
For individual consultation or questions about our products, call
1-800-628-0997